Alopecia Areata Dallas
Alopecia Areata Information
Alopecia Areata is a type of non-scarring hair loss (alopecia) that can have an unpredictable course and can be refractory at times to treatment intervention. Although the exact cause is unknown, the thought is that AA is a condition in which T lymphocytes attack one’s own hair follicles. The clinical presentation of Alopecia Areata (details to follow) involve anywhere from patchy oval/circular losses of hair to complete absence of scalp and even body hair. In most cases of patchy Alopecia Areata, resolution is complete within one year without treatment. In general, treatment is not curative but palliative only.
Alopecia Areata Causes
Epidemiology
Alopecia Areata can occur at any age but approximately 50% appear before the age of 20. The average lifetime risk is 1.7%. Conflicting reports have been given as to whether a higher number of men or women are affected by Alopecia Areata.
Genetics
There is a significant positive family history in those with Alopecia Areata, ranging from 10 to 42% of cases. Several genes, such as the human leukocyte antigen (HLA) located on Chromosome 6 has been linked to Alopecia Areata. Alopecia Areata has also been found to be more prevalent in individuals with Down’s syndrome and Turner syndrome. Given an 8.8% increased frequency of AA in Down’s syndrome the thought is that a gene location on Chromosome 21 may be partly responsible for Alopecia Areata. Most likely, Alopecia Areata has both a polygenic component and an environmental trigger as inciting factors.
Disease Associations
Allergic rhinitis, asthma, and atopic dermatitis have been associated with Alopecia Areata. In some studies, atopy was found to be present in more than 40% of Alopecia Areata Patients compared with 20% in the general population. Thyroid disease has also been found to be linked to Alopecia Areata. Alopecia Areata has also been found to be present in autoimmune type diseases like lupus, rheumatoid arthritis, myasthenia gravis, scleroderma, lichen planus, etc. HIV patients have also been shown to have a higher incidence of Alopecia Areata.
Immune Dysregulation
Alopecia Areata has been proposed to be an autoimmune disorder involving T-cell interaction with aberrant HLA-DR antigens expressed in hair follicle keratinocytes.
The Nervous System
Abnormal electroencephalographic (EEG) readings have been noted in Alopecia Areata patients suggesting a general neuronal defect. Patients with Alopecia Areata have also reported an increased incidence of itching, tingling, and slight pain when combing or touching the hair.
Emotional Stress
Although stressful life events and psychiatric disorders have been studied in their relationship with Alopecia Areata, the results of these studies have been conflicting.
Histology
On tissue diagnosis in an area of active Alopecia Areata, there is a peribulbar lymphocytic infiltrate (“swarm of bees”) without scarring. This is a classic finding in the active periphery of Alopecia Areata lesions. The inflammatory infiltrate is composed mainly of T-lymphocytes with macrophages and Langerhans cells. Miniaturization of the hair with numerous fibrous tracts and pigment incontinence are observed. There is a decreased anagen to telogen ratio.
Clinical Features
The characteristic lesion of Alopecia Areata is commonly round or oval, totally bald, smooth patch in the hair-bearing scalp. The patch can have a peach or pink color. Hair loss is noted to have both intact and fractured hairs. The fractured hairs have a broader distal (end) segment than near the scalp and are known as “exclamation point” hairs. As mentioned above, itchiness, tenderness, or pain can be associated in the lesion even before the appearance of the patch.
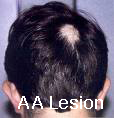
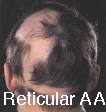
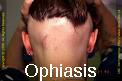
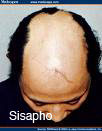
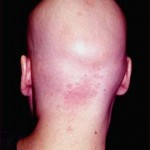
Types of Alopecia Areata by pattern:
- Patchy Alopecia Areata – with round or oval patches of hair loss (most common)
- Reticular Alopecia Areata– a reticulated pattern of patchy hair loss,
- Ophiasis Alopecia Areata– a band like Alopecia Areata that affects the temporo-occipital scalp,
- Ophiasis inversus (sisapho) – a rare band like pattern that affects the fronto-temporal scalp (opposite of ophiasis)
- Diffuse Alopecia Areata – affects the entire scalp.
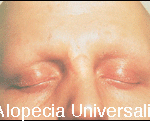
Alopecia Areata can also be categorized by the extent of involvement: 1) alopecia areata- partial loss of scalp hair, 2) alopecia totalis – 100% loss of scalp hair, 3) alopecia universalis – 100% loss of scalp hair plus body hair loss, e.g., eyebrows, eyelashes, beard, etc.
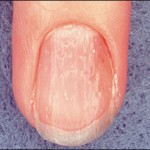 Nail dystrophy can be associated in 10 to 66% of cases of AA. Dystrophy may precede, coincide, or follow resolution of AA and include pitting, abnormal rows, abnormal grooves (Beau’s lines), etc.
Nail dystrophy can be associated in 10 to 66% of cases of AA. Dystrophy may precede, coincide, or follow resolution of AA and include pitting, abnormal rows, abnormal grooves (Beau’s lines), etc.
The progress of AA is unpredictable with the majority of individuals regrowing their hair entirely within one year without treatment. However, 7 to 10% can have a chronic form with poor prognosis related to atopy, presence of other immune diseases, positive family history of AA, young age of onset, nail dystrophy, and extensive hair loss.
Alopecia Areata Treatment
 At present, all treatments are to help reduce the signs and symptoms and are not curative. All local treatments can help treat the expression of the treated area but not help prevent any spread of the disease. Owing to the presence of T-cell immune dysregulation as the principal pathology, immune modulation remains the principal method to treat Alopecia Areata.
At present, all treatments are to help reduce the signs and symptoms and are not curative. All local treatments can help treat the expression of the treated area but not help prevent any spread of the disease. Owing to the presence of T-cell immune dysregulation as the principal pathology, immune modulation remains the principal method to treat Alopecia Areata.
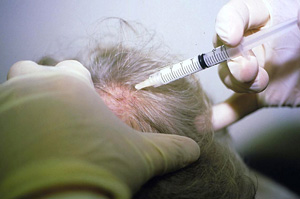
Intralesional steroid injections is the first-line therapy to treat adult patients with less than 50% of scalp involvement. 5 to 10 mg/cc of steroid is injected into the dermis in small aliquots of 0.1 cc with typical emergence of hair noted approximately 2 to 6 weeks after treatment. Treatment can be repeated every 1 to 2 weeks for a series of 3 rounds. Despite growth of hair, substantial percentage of those treated lose their regrowth after 12 weeks. Care should be taken to inject the steroid intradermally and not subcutaneously for risk of dermal atrophy.
Systemic oral corticosteroids are controversial in the treatment of AA and are not routinely used given their higher side-effect profile but can be useful in those individuals rapidly progressing to alopecia totalis. Cyclosporine has also been used similarly but with limited acceptance given the high relapse rate and unacceptable side effect profile.
Minoxidil 5%has been shown to provide cosmetically acceptable hair growth in 40% of patients with Alopecia Areata and should be thought of as a standard treatment option combined with intralesional steroid injections. However, Minoxidil is not helpful in patients with alopecia totalis or alopecia universalis.
Anthralin can be used in combination with minoxidil or by itself. Anthralin provides non-specific immune modulating effects, as it does in psoriasis. Cosmetically acceptable regrowth has been noted in 20 to 25% of cases of patchy Alopecia Areata. Anthralin 0.5 to 1.0% cream is applied daily, leaving it on the scalp for 20 to 30 minutes daily for 2 weeks then 45 minutes daily for 2 weeks up to a maximum of 1 hour. It should not be used on eyebrows or the beard. New hair growth is seen usually after 3 months of use.
Topical immunotherapy is the most effective treatment for chronic severe Alopecia Areata. Three agents used most often are dinitrochlorobenzene (DNCB), squaric acid dibutyl ester (SADBE), and diphenylcyclopropenone (DPCP). For adults with greater than 50% of hair loss from Alopecia Areata, these types of treatments may provide the most effective solution but still must be explained in great detail in terms of safety (toxicity), side effects, and efficacy. Treatment is carried out with a cotton swab over one half of the scalp in two directions until an allergic contact dermatitis arises. Scalp hair is not washed for 48 hours following treatment. The other side of the scalp is treated once hair growth is confirmed on the treated side. Once efficacy has been established, treatment can be progressively scaled back as far as interval.
Photochemotherapy with PUVA (Psoralen plus UVA light) has shown to be effective in modulating the abnormal T cell function and antigen presentation. Treatments are administered 2 to 3 times per week with gradual increase in UVA dosage. Unfortunately, there is a high relapse rate following cessation of PUVA intervention. Further, concerns that certain types of skin cancer like melanoma can be promoted with PUVA therapy have made it less than ideal.




